In a new research paper published in Oncotargetresearchers report an updated analysis from a phase I study of the spleen tyrosine kinase (SYK) and FMS-like tyrosine kinase 3 inhibitor mivavotinib. They present data for the overall cohort of lymphoma patients and the subgroup of patients with diffuse large B-cell lymphoma (DLBCL), including an expanded cohort not included in the initial report.
“Mivavotinib (TAK-659/CB-659) is an investigational, oral, reversible, potent dual inhibitor of spleen tyrosine kinase (SYK) and FMS-like tyrosine kinase 3 (FLT3). SYK is an essential component of the B-cell receptor signaling pathway; abnormal SYK signaling has been implicated in the pathogenesis of DLBCL and several other B-cell malignancies,” say the researchers.
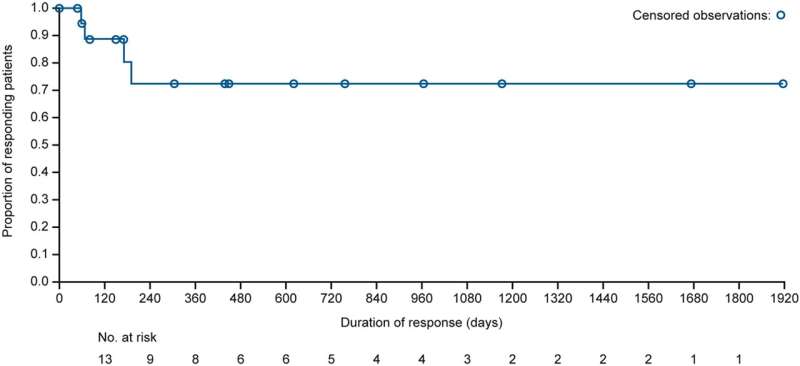
Patients with relapsed/refractory lymphoma for which no standard treatment was available received mivavotinib 60–120 mg once daily in 28-day cycles until disease progression/unacceptable toxicity. A total of 124 patients with lymphoma, including 89 with DLBCL, were enrolled. Overall response rates (ORR) in response-evaluable patients were 45% (43/95) and 38% (26/69), respectively. Median duration of response was 28.1 months overall and not reached in DLBCL responders.
In subgroups with DLBCL of germinal center B-cell (GCB) and non-GCB origin, ORR was 28% (11/40) and 58% (7/12), respectively. Median progression free survival was 2.0 and 1.6 months in the lymphoma and DLBCL cohorts, respectively. Grade ≥3 treatment-emergent adverse events occurred in 96% of all lymphoma patientsmany of which were limited to asymptomatic laboratory abnormalities; the most common were increased amylase (29%), neutropenia (27%), and hypophosphatemia (26%).
“These findings support SYK as a potential therapeutic target for the treatment of patients with B-cell lymphomas, including DLBCL,” note the researchers.
More information: Leo I. Gordon et al, Spleen tyrosine kinase/FMS-like tyrosine kinase-3 inhibition in relapsed/refractory B-cell lymphoma, including diffuse large B-cell lymphoma: updated data with mivavotinib (TAK-659/CB-659), Oncotarget (2023). DOI: 10.18632/oncotarget.28352
Provided byImpact Journals LLC
Citation: Mivavotinib for relapsed/refractory B-cell lymphoma (2023, February 1) retrieved 6 March 2023 from https://medicalxpress.com/news/2023-02-mivavotinib-relapsedrefractory-b-cell-lymphoma.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without the written permission. The content is provided for information purposes only.
Note: This article have been indexed to our site. We do not claim legitimacy, ownership or copyright of any of the content above. To see the article at original source Click Here